Regenerative

![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [1cc:2x0.5cc]](//www.osseoshop.com/cdn/shop/files/WebsitePictures-osbone_7.png?v=1709758039&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [1cc:2x0.5cc]](//www.osseoshop.com/cdn/shop/files/SyringeInstruction_62b83af4-cc05-4acf-857c-c8f03db5980d.png?v=1716321278&width=645)
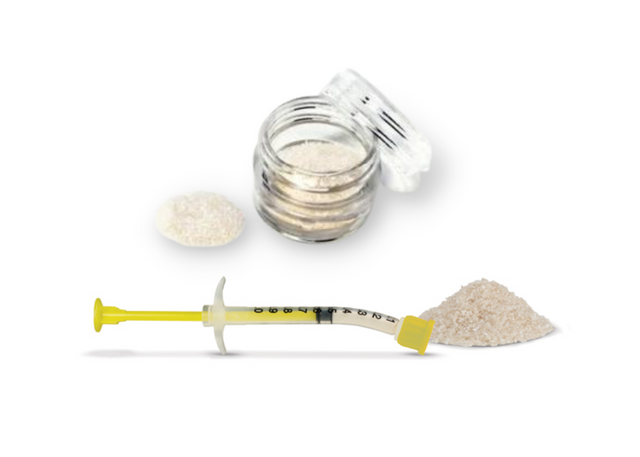
OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [1cc:2x0.5cc]
Mineralized Cortico-Cancellous (50/50) Bone Graft Prefilled in Curved Syringe, 250-1,000 microns OsseoSeal bone in syringe is a mixture of allograft mineralized cortical and cancellous powder in a convenient syringe with a filter cap! Curved syringe for better access to site. The cancellous and cortical blend forms an osteoconductive scaffold providing volume enhancement and effective site development for successful dental implant placement. Characteristics 50% Allograft Cortical Bone and 50% Cancellous Bone Sourced from Allosource tissue bank in the US Rigorous donor screening Low dose gamma irradiation
$85.00
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.5cc]](//www.osseoshop.com/cdn/shop/files/Main-0.5cc_1.png?v=1716321251&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.5cc]](//www.osseoshop.com/cdn/shop/files/Main-0.30.5_d32ebe9e-c64e-43c2-80b6-e4f658d86479.png?v=1716321251&width=645)
OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.5cc]
Mineralized Cortico-Cancellous (50/50) Bone Graft Prefilled in Curved Syringe, 250-1000 microns OsseoSeal bone in syringe is a mixture of allograft mineralized cortical and cancellous powder in a convenient syringe with a filter cap! Curved syringe for better access to site. The cancellous and cortical blend forms an osteoconductive scaffold providing volume enhancement and effective site development for successful dental implant placement. Characteristics 50% Allograft Cortical Bone and 50% Cancellous Bone Sourced from Allosource tissue bank in the US Rigorous donor screening Low dose gamma irradiation
$60.00
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.3cc]](//www.osseoshop.com/cdn/shop/files/Main-0.3cc_1.png?v=1716321186&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.3cc]](//www.osseoshop.com/cdn/shop/files/Main-0.30.5.png?v=1716321186&width=645)
OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.3cc]
Mineralized Cortico-Cancellous (50/50) Bone Graft Prefilled in Curved Syringe, 250-1000 microns OsseoSeal bone in syringe is a mixture of allograft mineralized cortical and cancellous powder in a convenient syringe with a filter cap! Curved syringe for better access to site. The cancellous and cortical blend forms an osteoconductive scaffold providing volume enhancement and effective site development for successful dental implant placement. Characteristics 50% Allograft Cortical Bone and 50% Cancellous Bone Sourced from Allosource tissue bank in the US Rigorous donor screening Low dose gamma irradiation
$40.00


OsteoGen Plug, Large (10x20mm), 10/pk: Get $50 off promo
*Effective Feburary 1, 2026: Price will increase from $549 to $579 The OsteoGen ® Plug combines bone graft with a collagen plug to yield the easiest and most affordable way to clinically deliver bone graft for socket preservation and ridge maintenance without the use of a membrane. Eliminate hassle of mixing particulate grafts. Eliminate graft wash out. Radiolucent on day of surgery – Radiopaque in 3-6 months Sold in packs packs of 10. Available in Large: 10mm in Diameter x 20mm in Length OsteoGen® proven safe, and clinically effective for use with implants for over 30 years. The OsteoGen® Bone Grafting Plug is the easiest and most affordable way to clinically deliver bone graft for ridge maintenance and socket preservation. The idea is simple - we take a collagen plug and fill it with our OsteoGen® non-ceramic bone graft crystals to create the OsteoGen® Bone Grafting Plug. The result is a bone graft combined with a collagen plug for ease of clinical delivery – all at the introductory special price of only $50 per extraction without the need for a membrane. The OsteoGen® Bone Grafting Plug combines our OsteoGen® Bioactive Resorbable Calcium Apatite with a bovine achilles tendon collagen matrix to create a structure that mimics the organic and inorganic components of physiologic bone. OsteoGen® is a bioactive and resorbable calcium apatite based bone graft that is physicochemically and crystallographically similar to human bone.1 The OsteoGen® non-ceramic production process yields a resorbable bone graft with a unique Ca:P ratio that is NOT a ß-TCP and NOT a non-resorbable dense ceramic HA, nor is it a biphasic mixture of the two.
$579.00


Resorbable Porcine Collagen Membrane
OsseoSeal™ is a resorbable collagen membrane derived from purified porcine pericardium. Once hydrated, the OsseoSeal™ collagen membrane has excellent tensile strength and is not easily torn. This ensures that clinicians can easily stretch the membrane without tearing. OsseoSeal™ the ideal membrane for tenting and other GBR procedures. Material: Type 1 porcine pericardium Resorption time: 3-4 months Fast hydration and excellent tensile strength Good adaptation to various defects Excellent tear function and duration One membrane in a box OsseoSeal is substantially equivalent to Bio-Guide. The primary difference between OssGuide and Bio-Gide is that OssGuide is manufactured from Porcine pericardium, while Bio-Gide is made from Porcine peritoneum.
$60.00 - $135.00


Titanium Mesh Membranes
Ti-matrix is placed after taking enough flap and then the bone grafting is performed afterwards, which ensures long-term stability in terms of flap healing. Ti-matrix ensures space maintenance for bone defect Ti-matrix is placed after taking enough flap and then the bone grafting is performed afterwards, which ensures long-term stability in terms of flap healing. Therefore, the right amounts of bone graft materials can be applied for primary closure as it was originally planned. Ti-matrix holds still even with one or two fixation screws. Ti-matrix can be used easily during surgery as it will enable surgeons to observe Made in Korea
$60.00 - $80.00


Non-Resorbable PTFE Membrane
Cytoflex Textured Tefguard is a non-resorbable PTFE membrane made of a proprietary micro-porous expanded polytetrafluoroethylene (ePTFE) material, that is ideal for socket preservations and bone grafting procedures. The textured version of Cytoflex Tefguard has superficial macro texture overlapping the micro pore texture on both surfaces, providing additional grips for flap attachment. Overlaying macro and micro textures on both surfaces! Allows nutrient permeation across the membrane. Resists fibroblasts and blocks bacterial penetration. Excellent Handling & Rigidity. Easily adaptable. Easily retrievable as one piece. Enhanced flap grips with fewer flap dehiscences Facilitates tissue regeneration despite flap recessions, or incomplete primary closure. Available in 12mmx24mm and 25mmx30mm. Provided double pouched and sterile. Cytoflex Textured Tefguard Benefits The textured version of Cytoflex® Tefguard® has superficial macro texture overlapping the micro pore texture on both surfaces. The cellular level micro texture is invisible to the naked eye, but can be seen at high magnifications. The macro texture provides additional grips for flap attachment. Allows nutrient permeation across the membrane for healthy bone regeneration. Resists fibroblasts and blocks bacterial penetration keeping the site safe from infection. Excellent Handling & Rigidity. Easily adaptable. Easily retrievable as one piece Better host tissue attachment with fewer flap dehiscences Easily retrievable as one piece Better host tissue attachment with fewer flap dehiscences Facilitates tissue regeneration despite flap recessions, or incomplete primary closure. Cytoflex ® Tefguard ® - Clinical Case Review MINIMALLY INVASIVE IMPLANT SITE GRAFTING TECHNIQUE Jenchun Chen DDS This is a 38 year-old female who presented with a crown-root fracture of the mandibular first molar and a thin gingival biotype. An immediate implant placement following tooth extraction was planned. A flapless, minimally invasive extraction and implant placement combined with guided tissue regeneration was employed to minimize soft and hard tissue recession. The tooth root was extracted with an intrasucular incision and a periosteal elevator. The extraction socket was curetted to remove all soft tissue remnants. After an implant was placed into the extraction site, the gap between the implant and the socket wall was filled with bone graft particles (Figures 1 & 2). A Tefguard® ePTFE membrane was trimmed to extend 3 mm beyond the socket walls and then tucked subperiosteally under the lingual flap, the buccal flap and underneath the interdental papilla using a curette. The membrane was allowed to rest passively over the socket (Figure 3), and was stabilized with a criss-cross absorbable PGA monofilament suture without primary closure (Figure 4). After one-week post operation, the graft site was uneventful, and the suture was removed (Figure 5). At three-week post-operation, the soft tissue overlying the exposed membrane demonstrated healing without signs of inflammation. An inadvertent fold in the membrane (introduced during membrane placement) was found at the distal buccal corner (Figure 6). The decision was made to remove the membrane early to prevent potential complications as a result of the folding of the membrane. After applying topical anesthetic, the membrane was easily removed by grasping with a tissue forcep. A dense, vascular connective tissue matrix was found underlying the membrane in the extraction socket upon membrane removal. Figure 7 shows the site at one week after membrane removal. Following membrane removal, keratinized gingiva began to form over the grafted socket. At six-week post-operation, the soft tissue was stable with preserved interproximal papillae and natural mucogingival architecture (Figure 8). This case demonstrates the use of a less invasive grafting technique using a micro porous ePTFE barrier.
$45.00 - $55.00


$50.00


Ora-Aid: Oral Bandage
15X25mm, 20pc/box Scroll down to watch instructional video Ora-Aid is a new concept pasting intra-oral patch to protect the affected area such as post implant, extraction, orthodontic, and ulcers. It is easy to use, just peel off and stick on the site! You can improve the predictability and success rate of intra oral surgery by good sealing and extra protection How to Use 1. Irrigate the wound with sterile or saline solution 2. Cut Ora-Aid into a proper shape and size. It should be trimmed according to the length of surgical site. (watch our videos for a demonstration of a cutting technique) 3. Slightly remove moisture, and excess saliva with gauze on the wound. Excessive moisture or exudation may reduce the adhesive strength and attachment time of Ora-Aid. 4. Remove transparent release paper and apply Ora-Aid on the wound. 5. Gently press Ora-Aid for 5 to 10 seconds while Ora-Aid adheres to wound. Click here for e-Catalog Research article Effects of a Topically Applied Oral Wound Dressing Film on Intra-oral Wound Healing in Rabbits Watch More Video (Click link below) Immediate implant placement after GBR Non-incision implantation after GBR Clinical Cases Suture Protocol1 How to attach Ora-Aid properly Socket Grafting Fixture removal and Bone Grafting Lateral Sinus Lift with implant placement One Stage Implant placement Clinical case Dr. Baranes 1 Clinical case Dr. Baranes 2
$92.00


UNIFY® PTFE Surgical Sutures 3.0
Item #:M-F318R19 Thread Size:3/0 Needle Spec:19mm, Reverse Cutting Comparable Needles:FS-2 / C-13 / C6 Thread Length:18 in (45cm) Color:White Packing:Box of 12 Pieces UNIFY® PTFE Surgical Sutures AD Surgical PTFE Sutures are the ideal suture for plastic, cosmetic and oral surgery Similar configurations to discontinued Gore-Tex® suture Very smooth surface, resulting in minimal tissue trauma Little or no package memory, soft handling, and knots securely Maintains tensile strength, perfect for suspension procedures Soft and flexible for patients (not stiff like most monofilaments) Biologically inert and chemically non-reactive Ultra sharp needle point for atraumatic tissue penetration Needle coated with silicone for smooth tissue passage Thread type: Monofilament White, Uncoated Strength duration: life time Absorb duration: N/A
$65.50


UNIFY® PTFE Surgical Sutures 4.0
Item #:M-F418R16 Thread Size:4/0 Needle Spec:16mm, Reverse Cutting Comparable Needles:FS-3 / C-12 / C22 Thread Length:18 in (45cm) Color:White Packing:Box of 12 Pieces UNIFY® PTFE Surgical Sutures AD Surgical PTFE Sutures are the ideal suture for plastic, cosmetic and oral surgery Similar configurations to discontinued Gore-Tex® suture Very smooth surface, resulting in minimal tissue trauma Little or no package memory, soft handling, and knots securely Maintains tensile strength, perfect for suspension procedures Soft and flexible for patients (not stiff like most monofilaments) Biologically inert and chemically non-reactive Ultra sharp needle point for atraumatic tissue penetration Needle coated with silicone for smooth tissue passage Thread type: Monofilament White, Uncoated Strength duration: life time Absorb duration: N/A
$65.50
![OsseoSeal Mineralized Allograft Cortical/Cancellous (50/50) Powder, 250-1000um [5cc:two packs of 2.5cc]](//www.osseoshop.com/cdn/shop/files/5cc_2x2.5ccjars_6.png?v=1709756716&width=645)
OsseoSeal Mineralized Allograft Cortical/Cancellous (50/50) Powder, 250-1000um [5cc:two packs of 2.5cc]
OsseoSeal / ExoGraft Allograft Cortical-Cancellous is a Freeze Dried Bone Allograft (FDBA) composed of cortical and cancellous bone. It has optimized porosity of cancellous bone and compactness of cortical bone to provide the best optimal scaffold and structural support for new bone formation. Our Allograft have been processed and sterilized to the highest standards and quality set forth by both American Association of Tissue Banks (AATB) and FDA. Characteristics 50% Allograft Cortical Bone and 50% Cancellous Bone 3-Dimensional lattice structure enables a fast revasculation Excellent Biocompatibility Osteoinduction Osteoconduction
$185.00
![OsseoSeal Mineralized Allograft Cortical/Cancellous, 250-1000um [2.5cc]](//www.osseoshop.com/cdn/shop/files/5cc_2x2.5ccjars_5_e5b8e887-5c70-4a6b-b9e1-f654f5b8a1fa.png?v=1709756425&width=645)
OsseoSeal Mineralized Allograft Cortical/Cancellous, 250-1000um [2.5cc]
OsseoSeal / ExoGraft Allograft Cortical-Cancellous is a Freeze Dried Bone Allograft (FDBA) composed of cortical and cancellous bone. It has optimized porosity of cancellous bone and compactness of cortical bone to provide the best optimal scaffold and structural support for new bone formation. Our Allograft have been processed and sterilized to the highest standards and quality set forth by both American Association of Tissue Banks (AATB) and FDA. Characteristics 50% Allograft Cortical Bone and 50% Cancellous Bone 3-Dimensional lattice structure enables a fast revasculation Excellent Biocompatibility Osteoinduction Osteoconduction
$100.00


Curagen™ Collagen Wound Dressing Plug, 10x20mm (3/8" x 3/4"), 10 /box
Curagen™ is a porous and absorbent collagen wound dressing. It isesigned for use in tooth extraction sites and oral surgeries to stop bleeding and reduce post operative pain. Curagen™ wound dressing is made of non-crosslinked bovine Achilles tendon collagen and available in various sizes and configurations. Large collagen pore structure 4-week long resorption duration Lessen post-surgical pain & duration Higher bone mineralization density Ideal for Extraction sites, Oral surgery, Donor sites Heliplug Alternative
$90.00
![OsseoSeal Xenograft Bovine Powder, 250-1000um [0.5cc]](//www.osseoshop.com/cdn/shop/files/1_6f4cbcb4-bc26-4679-8173-5f2003904ef3.png?v=1775499665&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [0.5cc]](//www.osseoshop.com/cdn/shop/files/OSBovinebone_2_1296x_bcaf5946-5930-4fd2-9398-733037a6b63f.png?v=1775499679&width=645)
OsseoSeal Xenograft Bovine Powder, 250-1000um [0.5cc]
OsseoSeal Xenograft Bovine Powder, 250-1000um Chemically and structurally comparable to mineralized cancellous human bone The highly purified osteoconductive mineralstructure is obtained from natural bovine bone through a manufacturing process that guarantees a 96% phase purity in strict compliance with international standards. Manufacturer: Odontit for Synergy Bone Matrix material, private labeled under name OsseoSeal.
$35.00 - $175.00
![OsseoSeal Xenograft Bovine Powder, 250-1000um [1cc]](//www.osseoshop.com/cdn/shop/files/2_7c312d35-56a2-489a-b197-3635dd2a350b.png?v=1775501693&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [1cc]](//www.osseoshop.com/cdn/shop/files/OSBovinebone_2_1296x_4a845c12-e139-459b-b18c-f5685264e17f.png?v=1775501939&width=645)
OsseoSeal Xenograft Bovine Powder, 250-1000um [1cc]
OsseoSeal Xenograft Bovine Powder, 250-1000um Chemically and structurally comparable to mineralized cancellous human bone The highly purified osteoconductive mineralstructure is obtained from natural bovine bone through a manufacturing process that guarantees a 96% phase purity in strict compliance with international standards. Manufacturer: Odontit for Synergy Bone Matrix material, private labeled under name OsseoSeal.
$50.00 - $250.00
![OsseoSeal Xenograft Bovine Powder, 250-1000um [4cc]](//www.osseoshop.com/cdn/shop/files/3_3b770848-5016-4485-8c26-6c25f849deaf.png?v=1775501939&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [4cc]](//www.osseoshop.com/cdn/shop/files/OSBovinebone_2_1296x_4a845c12-e139-459b-b18c-f5685264e17f.png?v=1775501939&width=645)
OsseoSeal Xenograft Bovine Powder, 250-1000um [4cc]
OsseoSeal Xenograft Bovine Powder, 250-1000um Chemically and structurally comparable to mineralized cancellous human bone The highly purified osteoconductive mineralstructure is obtained from natural bovine bone through a manufacturing process that guarantees a 96% phase purity in strict compliance with international standards. Manufacturer: Odontit for Synergy Bone Matrix material, private labeled under name OsseoSeal.
$120.00 - $600.00

![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [1cc:2x0.5cc]](http://www.osseoshop.com/cdn/shop/files/WebsitePictures-osbone_7.png?v=1709758039&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [1cc:2x0.5cc]](http://www.osseoshop.com/cdn/shop/files/SyringeInstruction_62b83af4-cc05-4acf-857c-c8f03db5980d.png?v=1716321278&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.5cc]](http://www.osseoshop.com/cdn/shop/files/Main-0.5cc_1.png?v=1716321251&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.5cc]](http://www.osseoshop.com/cdn/shop/files/Main-0.30.5_d32ebe9e-c64e-43c2-80b6-e4f658d86479.png?v=1716321251&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.3cc]](http://www.osseoshop.com/cdn/shop/files/Main-0.3cc_1.png?v=1716321186&width=645)
![OsseoSeal Mineralized Cortico-Cancellous (50/50) Bone in Syringe [0.3cc]](http://www.osseoshop.com/cdn/shop/files/Main-0.30.5.png?v=1716321186&width=645)
















![OsseoSeal Mineralized Allograft Cortical/Cancellous (50/50) Powder, 250-1000um [5cc:two packs of 2.5cc]](http://www.osseoshop.com/cdn/shop/files/5cc_2x2.5ccjars_6.png?v=1709756716&width=645)
![OsseoSeal Mineralized Allograft Cortical/Cancellous, 250-1000um [2.5cc]](http://www.osseoshop.com/cdn/shop/files/5cc_2x2.5ccjars_5_e5b8e887-5c70-4a6b-b9e1-f654f5b8a1fa.png?v=1709756425&width=645)


![OsseoSeal Xenograft Bovine Powder, 250-1000um [0.5cc]](http://www.osseoshop.com/cdn/shop/files/1_6f4cbcb4-bc26-4679-8173-5f2003904ef3.png?v=1775499665&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [0.5cc]](http://www.osseoshop.com/cdn/shop/files/OSBovinebone_2_1296x_bcaf5946-5930-4fd2-9398-733037a6b63f.png?v=1775499679&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [1cc]](http://www.osseoshop.com/cdn/shop/files/2_7c312d35-56a2-489a-b197-3635dd2a350b.png?v=1775501693&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [1cc]](http://www.osseoshop.com/cdn/shop/files/OSBovinebone_2_1296x_4a845c12-e139-459b-b18c-f5685264e17f.png?v=1775501939&width=645)
![OsseoSeal Xenograft Bovine Powder, 250-1000um [4cc]](http://www.osseoshop.com/cdn/shop/files/3_3b770848-5016-4485-8c26-6c25f849deaf.png?v=1775501939&width=645)